Congratulations to Argha Chakraborty on his brilliant artwork on the cover of Plasma Processes and Polymers.

The artwork illustrates the concept for his recently published article.

Congratulations to Argha Chakraborty on his brilliant artwork on the cover of Plasma Processes and Polymers.

The artwork illustrates the concept for his recently published article.

Plasma-activated water’s potential contribution to ‘One Health’
Foodborne pathogens cause a major burden to public health and the economy, costing A$2.44 billion, and causing 48,000 hospitalisations annually in Australia. With an increasing global impact of foodborne illnesses and the emergence of antibiotic-resistant pathogens, new decontamination technologies should consider the One Health approach to human, animal and environmental health. This review explores the application of plasma-activated water (PAW) as a novel sanitisation method. We discuss the implications of adopting PAW as an environmentally friendly and cost-effective sanitiser through a multidisciplinary One Health perspective. The findings underscore the promising role of PAW in mitigating foodborne pathogens, offering a holistic solution that aligns with the principles of One Health for enhanced food safety and public health.

This review presents a summary of plastic films made from two abundant natural polymers, starch and chitosan. Films possess many useful attributes such as transparency, good physical strength, and barrier properties. Modifications are being investigated to improve the properties of the product such as reinforcement with nanoparticles, strengthening by cross-linking, and applying surface coatings to improve interfacial properties. We provide perspectives on the use of starch chitosan films as a biobased, biodegradable food packaging material. Additionally, a detailed life-cycle assessment compares the production of chitosan-based polymers to other bioplastics and petroleum-based alternatives. Finally, we predict which factors will be important in the future for making the production of chitosan films economically and environmentally sustainable.
I had the pleasure of contributing to a research article which describes the results of the Fungus Olympics competition that I previously wrote about.
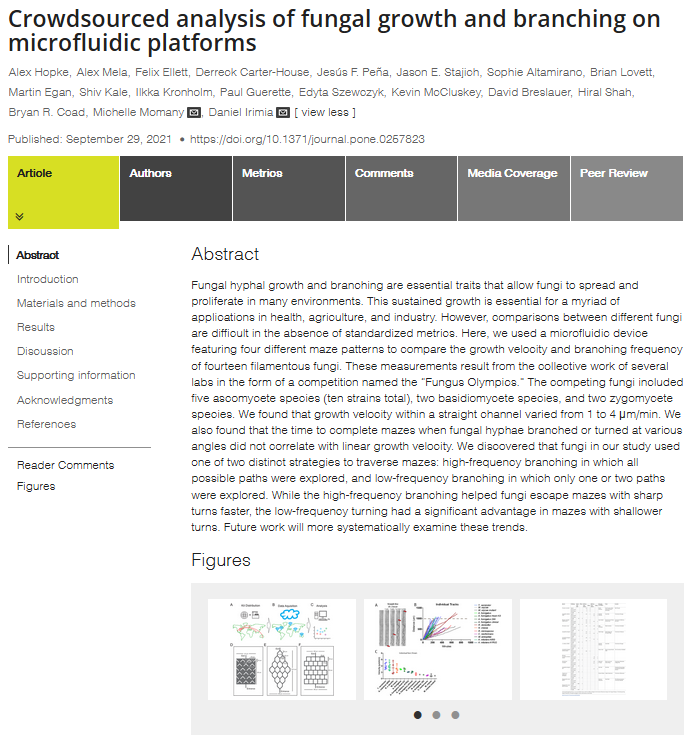
The article was published in PLoS ONE and can be found here.
https://doi.org/10.1371/journal.pone.0257823
Welcome to my webpage on my World Biomaterials Congress 2020 presentation:
The blind pathogen. Bioinspired and responsive materials for understanding the role of microbial surface sensing in the causes of plant and animal fungal diseases
This post contains information related to my oral presentation at the World Biomaterials Congress 2020.
Questions? Contact?
If you have questions or collaborative ideas, please get in touch.
Email: bryan.coad@adelaide.edu.au
Twitter DM: @DrBryanCoad
Additions & corrections:
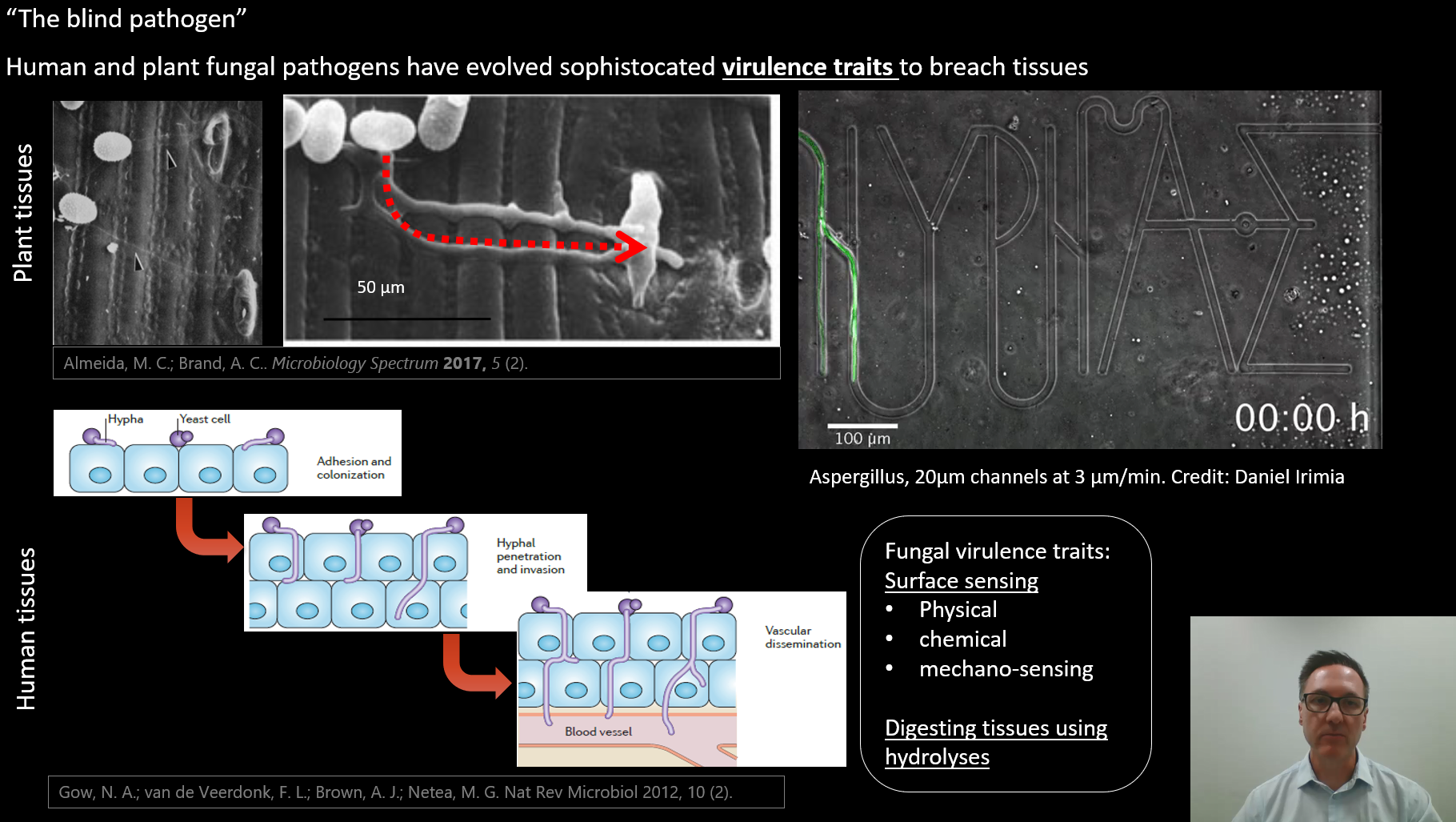
On slide 3 I showed the microfluidic device from Prof Daniel Irimia, but failed to give his affiliation. Prof Irimia is researcher and Deputy Director at Massachusetts General Hospital and Harvard Medical School, and organiser of the Fungus Olympics, an event in which my lab participated. You can read more about my involvement in the Fungus Olympics here.
References
Published paper on biodegradable materials:
Visualizing Biomaterial Degradation by Candida albicans Using Embedded Luminescent Molecules To Report on Substrate Digestion and Cellular Uptake of Hydrolysate. BR Coad; TD Michl; CA Bader; J Baranger; C Giles; GC Gonçalves; P Nath; SJ Lamont-Friedrich; M Johnsson; HJ Griesser; SE Plush. ACS Applied Bio Materials 2019, 2, 3934-3941. DOI: 10.1021/acsabm.9b00520
Reviews of fungi/plant relationships:
Cell Wall Responses to Biotrophic Fungal Pathogen Invasion. Annual Plant Reviews. J Chowdhury; BR Coad; A Little. 2019, 1001-1030. DOI: 10.1002/9781119312994.apr0634
Perspective on antifungal surface coatings for human health
Anti-infective Surface Coatings: Design and Therapeutic Promise against Device-Associated Infections. BR Coad; HJ Griesser; AY Peleg; A Traven. PLoS Pathogens 2016, 12, e1005598. DOI: 10.1371/journal.ppat.1005598
Information on this website:
Visualizing Biomaterial Degradation by Candida albicans Blog post on this paper
Blog post on the Fungus Olympics
Live cell imaging video of Blumeria graminis on a biomimetic surface coating
Site Navigation
I want to tell the stories behind some of my publications: why the work was done, who funded it, the roles of team members, or stumbling across unexpected results.
In this post, I’ll tell the story of the team effort behind our latest paper in the journal ACS Applied Bio Materials.
I’d like to highlight the great work from PhD student Javad Naderi.
Javad has published two papers on antifungal surface coatings.
The first, published in the Journal of Antimicrobial Chemotherapy, is on covalent coupling of two members of the echinocandin antifungal drug glass to surfaces. Both anidulafungin and micafungin surface coatings were highly antifungal vs Candida albicans.

Surface coatings with covalently attached anidulafungin and micafungin prevent Candida albicans biofilm formation. doi.org/10.1093/jac/dky437
However, perhaps the most important finding was that the anidulafungin surface killed yeast and prevented biofilm, and could be washed and rechallenged at least 5 times without losing efficacy.
This finding supports a mechanism of action where the drug is:
Along with previous publications on the other approved echinocandin, caspofungin, there is more evidence to suggest that the echinocandin drug class can be antifungal even when immobilised on surfaces (i.e. does not need diffusion to reach drug targets). Exactly how this happens is an active area or research for us.
The second explores the surface activity of another drug class, the polyenes. At first, it looks like drugs like amphotericin B remain active when attached to surfaces; however, if you are careful and you make sure to wash off ALL traces of loosely adsorbed compounds (leaving only contently attached) then this effect is eliminated.
Thus, not all surface coatings prepared using covalent attachment of antifungal drugs actually work.

Surface-grafted antimicrobial drugs: Possible misinterpretation of mechanism of action. doi.org/10.1116/1.5050043
Failure to account for compounds that can be washed off and possible misinterpretation of mechanism of action should be an issue that researchers are made aware of.
This article was a Featured Article and a Scilight published in the journal Biointerphases.
How can we improve medical implants so that they combat microbial colonization and reduce the problem of implant-associated infections?
Basic science approaches for the design antimicrobial surfaces and coatings has been reviewed but it is clear that attention for fungal pathogens has been missing.
Fungal pathogens are important because they
We have published a new review entitled The Importance of Fungal Pathogens and Antifungal Coatings in Medical Device Infections.
https://www.sciencedirect.com/science/article/pii/S0734975017301520

The review article is online now. Click the image to go to the website.
Highlights include:
This review is a significant addition to our 2014 review Biomaterials surfaces capable of resisting fungal attachment and biofilm formation. Biotech Adv. 2014; 32: 296-307.
This was a massive team effort that progressed over the course of one year. Our team of five authors wrote 17 pages of printed text and reviewed 89 references. This is the most comprehensive and up-to-date review in the field. We hope that the discussion on materials design will become an invaluable resource for researchers and feed into broader thinking about the wider field of design of antimicrobial biomaterials and polymicrobial biofilms.
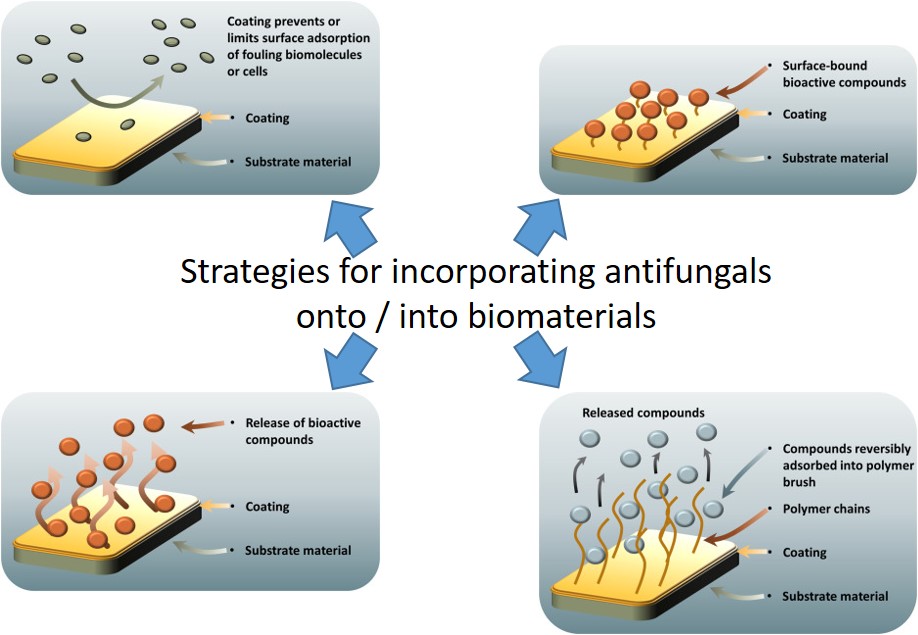
Materials strategies for incorporating antifungals onto and into biomaterials.
We have continued our investigation on coatings that could be used to combat deadly fungal infections. Implanted medical devices can become colonised by the fungal yeast Candida albicans despite sterilisation procedures. Therefore, a medical device with a surface modification that could kill Candida on contact may help to reduce infection and death from these infections.
Previously we have shown that the antifungal drug caspofungin can be attached to medical-grade plastics using an ultra-thin surface layer only 20 nm thick. Because this coating is thin and flat, the caspofungin molecules essentially form a flat layer (approximately a 2 dimensional surface).
Our latest research has investigated the possibility of instead attaching caspofungin on polymer brushes. This allows the drug to be presented on flexible polymer linkers, extending hundreds of nanometres from the surface, and would improve the way the drug interacts with the fungal cell wall.
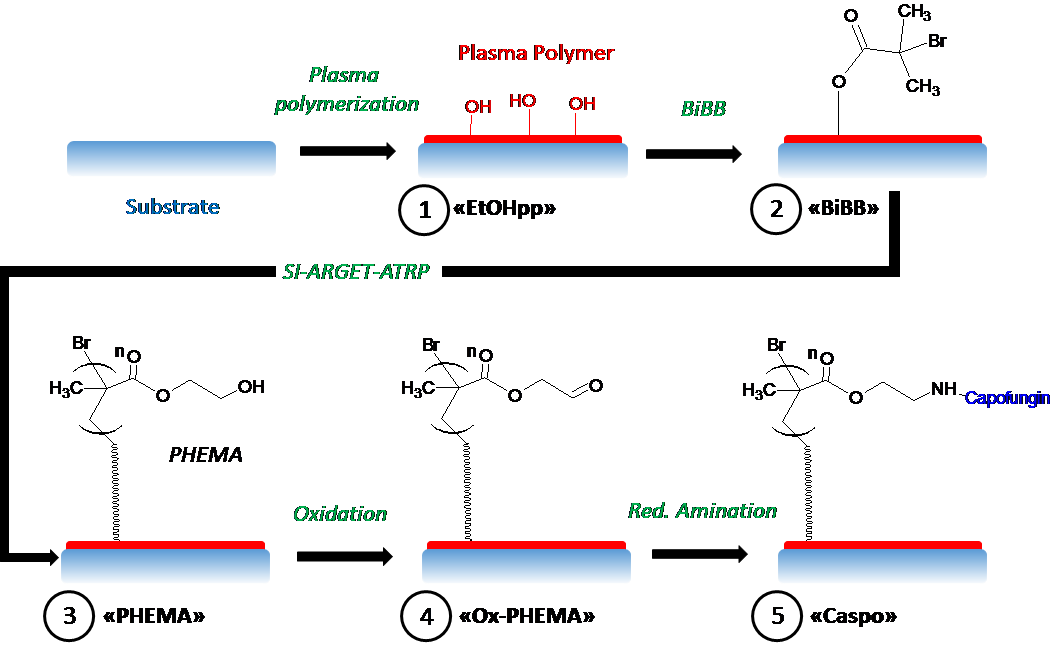
Depiction of the reaction steps, combining plasma polymerization and ARGET-ATRP to covalently attach amine-containing drugs, such as caspofungin, to surface-grafted PHEMA.
This is an important advancement in helping us to understand how surface-immobilised drugs interact with the fungal cell wall. We will use this information to understand how antibiotics work mecahanistically – that is, how tethered drugs would be able to penetrate into the cell wall and find their target of action which ends up killing the cell. Finding new mechanisms of action for antibiotic drugs could be one way that we can find new uses for a dwindling supply of effective antibiotic agents.
The work has been published in the journal Biointerphases and is an “Editor’s Pick”. This article is freely accessible through open access publishing.
We gratefully acknowledge the Australian Research Council for funding. (Discovery Project DP150101674. “Combating fungal biofilm growth on surfaces”.)