Hospital acquired infections are a public health crisis and a leading cause of death world-wide. In the USA over 200 people die each day from hospital acquired infections with over 80% of these related to contaminated surfaces of implanted medical devices. While there is a good level of understanding and research into bacterial infections, fungal infections are the hidden killers – often missed in diagnosis. Treatment with antifungal drugs is therefore delayed and often comes too late, with devastating consequences. Rapidly multiplying on surfaces in a protected slimy coating known as a biofilm, mature fungal infections can spread into the blood and are often more effective when compared to bacteria in host mortality — giving a less than 50/50 chance of patient survival.
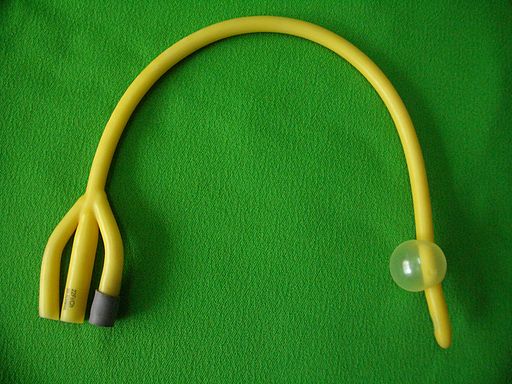
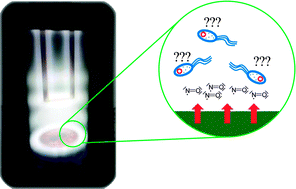
The surface of urinary catheters is frequently a site for bacterial and fungal biofilm growth and often leads to infection.
The understanding of how to fight fungal infections from surfaces will be given a major boost by a 4-year grant from the Australian Research Council’s Discovery Grant Program. Recognizing the need for cross disciplinary research at the nexus of physical chemistry, materials science, and biology, the team led by University of South Australia Researchers (the Mycology / Surface Interfaces Group) will bring together world-leading polymer chemists and mycologists across three countries (Australia, Switzerland and the UK) to drive breakthrough research into effective antifungal surface coatings.
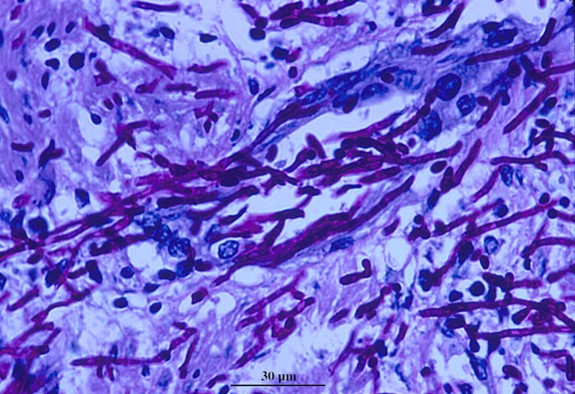
Candida albicans, common and mostly benign to healthy individuals, becomes deadly inside the body when it forms biofilms and invades tissues.
Professor Hans Griesser and Senior Research Fellow Dr. Bryan Coad of UniSA’s Mawson Institute will lead the grant. “This grant will bring Australian discoveries to the forefront in what is a new research area,” says Dr. Coad. “Strengthened by the expertise from our international partners, we see potential to deliver outcomes likely to resonate with our scientific peers the world over.”
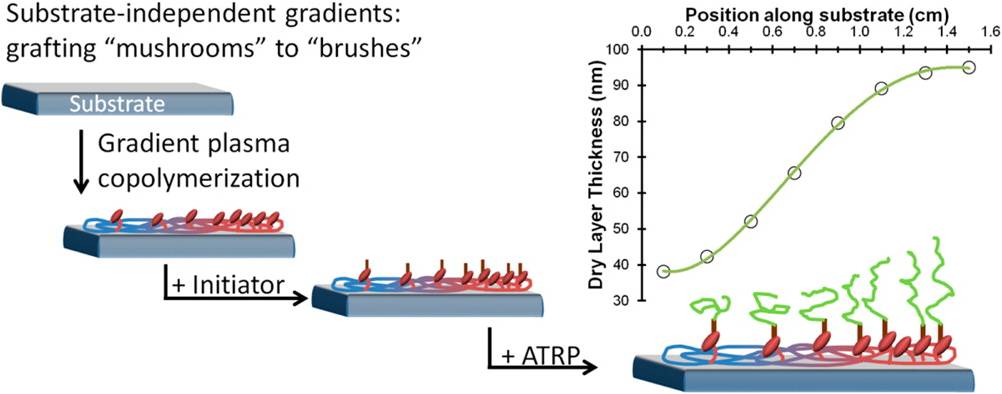
The four-year research program will develop advanced material surface coatings at UniSA and unravel the design principles needed to transform them into 3-D polymeric platforms in collaboration with partner investigator Prof. Harm-Anton Klok at EPFL Switzerland, an expert in bioactive polymer coatings. Direct visualization of fungal cells on surfaces using live cell imaging by partner investigator Prof. Nick Read at the University of Manchester (Manchester Fungal Infection Group) will reveal the fate of cells and provide direct evidence for the mechanisms of action. “The team will tie the physical and chemical properties of their surface coatings to their specific biological consequences. For fungi, this has never been done before so there is fantastic opportunity to make fundamental discoveries,” says Dr. Coad.